Alkane

In organic chemistry, an alkane, or paraffin (a historical trivial name that also has other meanings), is an acyclic saturated hydrocarbon. In other words, an alkane consists of hydrogen and carbon atoms arranged in a tree structure in which all the carbon–carbon bonds are single.[1] Alkanes have the general chemical formula CnH2n+2. The alkanes range in complexity from the simplest case of methane (CH4), where n = 1 (sometimes called the parent molecule), to arbitrarily large and complex molecules, like pentacontane (C50H102) or 6-ethyl-2-methyl-5-(1-methylethyl) octane, an isomer of tetradecane (C14H30).
The International Union of Pure and Applied Chemistry (IUPAC) defines alkanes as "acyclic branched or unbranched hydrocarbons having the general formula CnH2n+2, and therefore consisting entirely of hydrogen atoms and saturated carbon atoms". However, some sources use the term to denote any saturated hydrocarbon, including those that are either monocyclic (i.e. the cycloalkanes) or polycyclic, despite them having a distinct general formula (e.g. cycloalkanes are CnH2n).
In an alkane, each carbon atom is sp3-hybridized with 4 sigma bonds (either C–C or C–H), and each hydrogen atom is joined to one of the carbon atoms (in a C–H bond). The longest series of linked carbon atoms in a molecule is known as its carbon skeleton or carbon backbone. The number of carbon atoms may be considered as the size of the alkane.
One group of the higher alkanes are waxes, solids at standard ambient temperature and pressure (SATP), for which the number of carbon atoms in the carbon backbone is greater than about 17. With their repeated –CH2 units, the alkanes constitute a homologous series of organic compounds in which the members differ in molecular mass by multiples of 14.03 u (the total mass of each such methylene-bridge unit, which comprises a single carbon atom of mass 12.01 u and two hydrogen atoms of mass ~1.01 u each).
Methane is produced by methanogenic bacteria and some long-chain alkanes function as pheromones in certain animal species or as protective waxes in plants and fungi. Nevertheless, most alkanes do not have much biological activity. They can be viewed as molecular trees upon which can be hung the more active/reactive functional groups of biological molecules.
The alkanes have two main commercial sources: petroleum (crude oil) and natural gas.
An alkyl group is an alkane-based molecular fragment that bears one open valence for bonding. They are generally abbreviated with the symbol for any organyl group, R, although Alk is sometimes used to specifically symbolize an alkyl group (as opposed to an alkenyl group or aryl group).
Structure and classification
[edit]Ordinarily the C-C single bond distance is 1.53 ångströms (1.53×10−10 m).[2] Saturated hydrocarbons can be linear, branched, or cyclic. The third group is sometimes called cycloalkanes.[1] Very complicated structures are possible by combining linear, branch, cyclic alkanes.
Isomerism
[edit]
Alkanes with more than three carbon atoms can be arranged in various ways, forming structural isomers. The simplest isomer of an alkane is the one in which the carbon atoms are arranged in a single chain with no branches. This isomer is sometimes called the n-isomer (n for "normal", although it is not necessarily the most common). However, the chain of carbon atoms may also be branched at one or more points. The number of possible isomers increases rapidly with the number of carbon atoms. For example, for acyclic alkanes:[3]
- C1: methane only
- C2: ethane only
- C3: propane only
- C4: 2 isomers: butane and isobutane
- C5: 3 isomers: pentane, isopentane, and neopentane
- C6: 5 isomers: hexane, 2-methylpentane, 3-methylpentane, 2,2-dimethylbutane, and 2,3-dimethylbutane
- C7: 9 isomers: heptane, 2-methylhexane, 3-methylhexane, 2,2-dimethylpentane, 2,3-dimethylpentane, 2,4-dimethylpentane, 3,3-dimethylpentane, 3-ethylpentane, 2,2,3-trimethylbutane
- C8: 18 isomers: octane, 2-methylheptane, 3-methylheptane, 4-methylheptane, 2,2-dimethylhexane, 2,3-dimethylhexane, 2,4-dimethylhexane, 2,5-dimethylhexane, 3,3-dimethylhexane, 3,4-dimethylhexane, 3-ethylhexane, 2,2,3-trimethylpentane, 2,2,4-trimethylpentane, 2,3,3-trimethylpentane, 2,3,4-trimethylpentane, 3-ethyl-2-methylpentane, 3-ethyl-3-methylpentane, 2,2,3,3-tetramethylbutane
- C9: 35 isomers
- C10: 75 isomers
- C12: 355 isomers
- C32: 27,711,253,769 isomers
- C60: 22,158,734,535,770,411,074,184 isomers, many of which are not stable
Branched alkanes can be chiral. For example, 3-methylhexane and its higher homologues are chiral due to their stereogenic center at carbon atom number 3. The above list only includes differences of connectivity, not stereochemistry. In addition to the alkane isomers, the chain of carbon atoms may form one or more rings. Such compounds are called cycloalkanes, and are also excluded from the above list because changing the number of rings changes the molecular formula. For example, cyclobutane and methylcyclopropane are isomers of each other (C4H8), but are not isomers of butane (C4H10).
Branched alkanes are more thermodynamically stable than their linear (or less branched) isomers. For example, the highly branched 2,2,3,3-tetramethylbutane is about 1.9 kcal/mol more stable than its linear isomer, n-octane.[4]
Nomenclature
[edit]The IUPAC nomenclature (systematic way of naming compounds) for alkanes is based on identifying hydrocarbon chains. Unbranched, saturated hydrocarbon chains are named systematically with a Greek numerical prefix denoting the number of carbons and the suffix "-ane".[5]
In 1866, August Wilhelm von Hofmann suggested systematizing nomenclature by using the whole sequence of vowels a, e, i, o and u to create suffixes -ane, -ene, -ine (or -yne), -one, -une, for the hydrocarbons CnH2n+2, CnH2n, CnH2n−2, CnH2n−4, CnH2n−6.[6] In modern nomenclature, the first three specifically name hydrocarbons with single, double and triple bonds;[7] while "-one" now represents a ketone.
Linear alkanes
[edit]Straight-chain alkanes are sometimes indicated by the prefix "n-" or "n-"(for "normal") where a non-linear isomer exists. Although this is not strictly necessary and is not part of the IUPAC naming system, the usage is still common in cases where one wishes to emphasize or distinguish between the straight-chain and branched-chain isomers, e.g., "n-butane" rather than simply "butane" to differentiate it from isobutane. Alternative names for this group used in the petroleum industry are linear paraffins or n-paraffins.
The first eight members of the series (in terms of number of carbon atoms) are named as follows:
- methane
- CH4 – one carbon and 4 hydrogen
- ethane
- C2H6 – two carbon and 6 hydrogen
- propane
- C3H8 – three carbon and 8 hydrogen
- butane
- C4H10 – four carbon and 10 hydrogen
- pentane
- C5H12 – five carbon and 12 hydrogen
- hexane
- C6H14 – six carbon and 14 hydrogen
- heptane
- C7H16 – seven carbons and 16 hydrogen
- octane
- C8H18 – eight carbons and 18 hydrogen
The first four names were derived from methanol, ether, propionic acid and butyric acid. Alkanes with five or more carbon atoms are named by adding the suffix -ane to the appropriate numerical multiplier prefix[8] with elision of any terminal vowel (-a or -o) from the basic numerical term. Hence, pentane, C5H12; hexane, C6H14; heptane, C7H16; octane, C8H18; etc. The numeral prefix is generally Greek; however, alkanes with a carbon atom count ending in nine, for example nonane, use the Latin prefix non-.
Branched alkanes
[edit]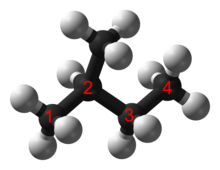
Simple branched alkanes often have a common name using a prefix to distinguish them from linear alkanes, for example n-pentane, isopentane, and neopentane.
IUPAC naming conventions can be used to produce a systematic name.
The key steps in the naming of more complicated branched alkanes are as follows:[9]
- Identify the longest continuous chain of carbon atoms
- Name this longest root chain using standard naming rules
- Name each side chain by changing the suffix of the name of the alkane from "-ane" to "-yl"
- Number the longest continuous chain in order to give the lowest possible numbers for the side-chains[10]
- Number and name the side chains before the name of the root chain
- If there are multiple side chains of the same type, use prefixes such as "di-" and "tri-" to indicate it as such, and number each one.
- Add side chain names in alphabetical (disregarding "di-" etc. prefixes) order in front of the name of the root chain
| Common name | n-pentane | isopentane | neopentane |
|---|---|---|---|
| IUPAC name | pentane | 2-methylbutane | 2,2-dimethylpropane |
| Structure |  |
|
Saturated cyclic hydrocarbons
[edit]Though technically distinct from the alkanes, this class of hydrocarbons is referred to by some as the "cyclic alkanes." As their description implies, they contain one or more rings.
Simple cycloalkanes have a prefix "cyclo-" to distinguish them from alkanes. Cycloalkanes are named as per their acyclic counterparts with respect to the number of carbon atoms in their backbones, e.g., cyclopentane (C5H10) is a cycloalkane with 5 carbon atoms just like pentane (C5H12), but they are joined up in a five-membered ring. In a similar manner, propane and cyclopropane, butane and cyclobutane, etc.
Substituted cycloalkanes are named similarly to substituted alkanes – the cycloalkane ring is stated, and the substituents are according to their position on the ring, with the numbering decided by the Cahn–Ingold–Prelog priority rules.[8]
Trivial/common names
[edit]The trivial (non-systematic) name for alkanes is 'paraffins'. Together, alkanes are known as the 'paraffin series'. Trivial names for compounds are usually historical artifacts. They were coined before the development of systematic names, and have been retained due to familiar usage in industry. Cycloalkanes are also called naphthenes.[11][12]
Branched-chain alkanes are called isoparaffins. "Paraffin" is a general term and often does not distinguish between pure compounds and mixtures of isomers, i.e., compounds of the same chemical formula, e.g., pentane and isopentane.
- In IUPAC
The following trivial names are retained in the IUPAC system:
- isobutane for 2-methylpropane
- isopentane for 2-methylbutane
- neopentane for 2,2-dimethylpropane.
- Non-IUPAC
Some non-IUPAC trivial names are occasionally used:
- cetane, for hexadecane
- cerane, for hexacosane[13]
Physical properties
[edit]All alkanes are colorless.[14][15] Alkanes with the lowest molecular weights are gases, those of intermediate molecular weight are liquids, and the heaviest are waxy solids.[16][17]
Table of alkanes
[edit]| Alkane | Formula | Boiling point[note 1] [°C] |
Melting point[note 1] [°C] |
Density[note 1] [kg/m3] (at 20 °C) |
Isomers[note 2] |
|---|---|---|---|---|---|
| Methane | CH4 | −162 | −182 | 0.656 (gas) | 1 |
| Ethane | C2H6 | −89 | −183 | 1.26 (gas) | 1 |
| Propane | C3H8 | −42 | −188 | 2.01 (gas) | 1 |
| Butane | C4H10 | 0 | −138 | 2.48 (gas) | 2 |
| Pentane | C5H12 | 36 | −130 | 626 (liquid) | 3 |
| Hexane | C6H14 | 69 | −95 | 659 (liquid) | 5 |
| Heptane | C7H16 | 98 | −91 | 684 (liquid) | 9 |
| Octane | C8H18 | 126 | −57 | 703 (liquid) | 18 |
| Nonane | C9H20 | 151 | −54 | 718 (liquid) | 35 |
| Decane | C10H22 | 174 | −30 | 730 (liquid) | 75 |
| Undecane | C11H24 | 196 | −26 | 740 (liquid) | 159 |
| Dodecane | C12H26 | 216 | −10 | 749 (liquid) | 355 |
| Tridecane | C13H28 | 235 | −5.4 | 756 (liquid) | 802 |
| Tetradecane | C14H30 | 253 | 5.9 | 763 (liquid) | 1858 |
| Pentadecane | C15H32 | 270 | 10 | 769 (liquid) | 4347 |
| Hexadecane | C16H34 | 287 | 18 | 773 (liquid) | 10,359 |
| Heptadecane | C17H36 | 303 | 22 | 777 (solid) | 24,894 |
| Octadecane | C18H38 | 317 | 28 | 781 (solid) | 60,523 |
| Nonadecane | C19H40 | 330 | 32 | 785 (solid) | 148,284 |
| Icosane | C20H42 | 343 | 37 | 789 (solid) | 366,319 |
| Triacontane | C30H62 | 450 | 66 | 810 (solid) | 4,111,846,763 |
| Tetracontane | C40H82 | 525 | 82 | 817 (solid) | 62,481,801,147,341 |
| Pentacontane | C50H102 | 575 | 91 | 824 (solid) | 1,117,743,651,746,953,270 |
| Hexacontane | C60H122 | 625 | 100 | 829 (solid) | 2.21587345357704×1022 |
| Heptacontane | C70H142 | 653 | 109 | 869 (solid) | 4.71484798515330×1026 |
| |||||
Boiling point
[edit]
Alkanes experience intermolecular van der Waals forces. The cumulative effects of these intermolecular forces give rise to greater boiling points of alkanes.[18]
Two factors influence the strength of the van der Waals forces:
- the number of electrons surrounding the molecule, which increases with the alkane's molecular weight
- the surface area of the molecule
Under standard conditions, from CH4 to C4H10 alkanes are gaseous; from C5H12 to C17H36 they are liquids; and after C18H38 they are solids. As the boiling point of alkanes is primarily determined by weight, it should not be a surprise that the boiling point has an almost linear relationship with the size (molecular weight) of the molecule. As a rule of thumb, the boiling point rises 20–30 °C for each carbon added to the chain; this rule applies to other homologous series.[18]
A straight-chain alkane will have a boiling point higher than a branched-chain alkane due to the greater surface area in contact, and thus greater van der Waals forces, between adjacent molecules. For example, compare isobutane (2-methylpropane) and n-butane (butane), which boil at −12 and 0 °C, and 2,2-dimethylbutane and 2,3-dimethylbutane which boil at 50 and 58 °C, respectively.[18]
On the other hand, cycloalkanes tend to have higher boiling points than their linear counterparts due to the locked conformations of the molecules, which give a plane of intermolecular contact.
Melting points
[edit]The melting points of the alkanes follow a similar trend to boiling points for the same reason as outlined above. That is, (all other things being equal) the larger the molecule the higher the melting point. There is one significant difference between boiling points and melting points. Solids have a more rigid and fixed structure than liquids. This rigid structure requires energy to break down. Thus the better put together solid structures will require more energy to break apart. For alkanes, this can be seen from the graph above (i.e., the blue line). The odd-numbered alkanes have a lower trend in melting points than even-numbered alkanes. This is because even-numbered alkanes pack well in the solid phase, forming a well-organized structure which requires more energy to break apart. The odd-numbered alkanes pack less well and so the "looser"-organized solid packing structure requires less energy to break apart.[19] For a visualization of the crystal structures see.[20]
The melting points of branched-chain alkanes can be either higher or lower than those of the corresponding straight-chain alkanes, again depending on the ability of the alkane in question to pack well in the solid phase.
Conductivity and solubility
[edit]Alkanes do not conduct electricity in any way, nor are they substantially polarized by an electric field. For this reason, they do not form hydrogen bonds and are insoluble in polar solvents such as water. Since the hydrogen bonds between individual water molecules are aligned away from an alkane molecule, the coexistence of an alkane and water leads to an increase in molecular order (a reduction in entropy). As there is no significant bonding between water molecules and alkane molecules, the second law of thermodynamics suggests that this reduction in entropy should be minimized by minimizing the contact between alkane and water: Alkanes are said to be hydrophobic as they are insoluble in water.
Their solubility in nonpolar solvents is relatively high, a property that is called lipophilicity. Alkanes are, for example, miscible in all proportions among themselves.
The density of the alkanes usually increases with the number of carbon atoms but remains less than that of water. Hence, alkanes form the upper layer in an alkane–water mixture.[21]
Molecular geometry
[edit]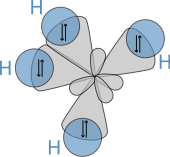
The molecular structure of the alkanes directly affects their physical and chemical characteristics. It is derived from the electron configuration of carbon, which has four valence electrons. The carbon atoms in alkanes are described as sp3 hybrids; that is to say that, to a good approximation, the valence electrons are in orbitals directed towards the corners of a tetrahedron which are derived from the combination of the 2s orbital and the three 2p orbitals. Geometrically, the angle between the bonds are cos−1(−1/3) ≈ 109.47°. This is exact for the case of methane, while larger alkanes containing a combination of C–H and C–C bonds generally have bonds that are within several degrees of this idealized value.
Bond lengths and bond angles
[edit]
An alkane has only C–H and C–C single bonds. The former result from the overlap of an sp3 orbital of carbon with the 1s orbital of a hydrogen; the latter by the overlap of two sp3 orbitals on adjacent carbon atoms. The bond lengths amount to 1.09 × 10−10 m for a C–H bond and 1.54 × 10−10 m for a C–C bond.
The spatial arrangement of the bonds is similar to that of the four sp3 orbitals—they are tetrahedrally arranged, with an angle of 109.47° between them. Structural formulae that represent the bonds as being at right angles to one another, while both common and useful, do not accurately depict the geometry.
Conformation
[edit]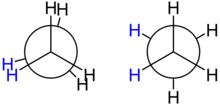

The spatial arrangement of the C-C and C-H bonds are described by the torsion angles of the molecule is known as its conformation. In ethane, the simplest case for studying the conformation of alkanes, there is nearly free rotation about a carbon–carbon single bond. Two limiting conformations are important: eclipsed conformation and staggered conformation. The staggered conformation is 12.6 kJ/mol (3.0 kcal/mol) lower in energy (more stable) than the eclipsed conformation (the least stable). In highly branched alkanes, the bond angle may differ from the optimal value (109.5°) to accommodate bulky groups. Such distortions introduce a tension in the molecule, known as steric hindrance or strain. Strain substantially increases reactivity.[22]
Spectroscopic properties
[edit]Spectroscopic signatures for alkanes are obtainable by the major characterization techniques.[23]
Infrared spectroscopy
[edit]The C-H stretching mode gives a strong absorptions between 2850 and 2960 cm−1 and weaker bands for the C-C stretching mode absorbs between 800 and 1300 cm−1. The carbon–hydrogen bending modes depend on the nature of the group: methyl groups show bands at 1450 cm−1 and 1375 cm−1, while methylene groups show bands at 1465 cm−1 and 1450 cm−1.[24] Carbon chains with more than four carbon atoms show a weak absorption at around 725 cm−1.
NMR spectroscopy
[edit]The proton resonances of alkanes are usually found at δH = 0.5–1.5. The carbon-13 resonances depend on the number of hydrogen atoms attached to the carbon: δC = 8–30 (primary, methyl, –CH3), 15–55 (secondary, methylene, –CH2–), 20–60 (tertiary, methyne, C–H) and quaternary. The carbon-13 resonance of quaternary carbon atoms is characteristically weak, due to the lack of nuclear Overhauser effect and the long relaxation time, and can be missed in weak samples, or samples that have not been run for a sufficiently long time.
Mass spectrometry
[edit]Since alkanes have high ionization energies, their electron impact mass spectra show weak currents for their molecular ions. The fragmentation pattern can be difficult to interpret, but in the case of branched chain alkanes, the carbon chain is preferentially cleaved at tertiary or quaternary carbons due to the relative stability of the resulting free radicals. The mass spectra for straight-chain alkanes is illustrated by that for dodecane: the fragment resulting from the loss of a single methyl group (M − 15) is absent, fragments are more intense than the molecular ion and are spaced by intervals of 14 mass units, corresponding to loss of CH2 groups.[25]
Chemical properties
[edit]Alkanes are only weakly reactive with most chemical compounds. They only reacts with the strongest of electrophilic reagents by virtue of their strong C–H bonds (~100 kcal/mol) and C–C bonds (~90 kcal/mol). They are also relatively unreactive toward free radicals. This inertness is the source of the term paraffins (with the meaning here of "lacking affinity"). In crude oil the alkane molecules have remained chemically unchanged for millions of years.
Acid-base behavior
[edit]The acid dissociation constant (pKa) values of all alkanes are estimated to range from 50 to 70, depending on the extrapolation method, hence they are extremely weak acids that are practically inert to bases (see: carbon acids). They are also extremely weak bases, undergoing no observable protonation in pure sulfuric acid (H0 ~ −12), although superacids that are at least millions of times stronger have been known to protonate them to give hypercoordinate alkanium ions (see: methanium ion). Thus, a mixture of antimony pentafluoride (SbF5) and fluorosulfonic acid (HSO3F), called magic acid, can protonate alkanes.[26]
Reactions with oxygen (combustion reaction)
[edit]All alkanes react with oxygen in a combustion reaction, although they become increasingly difficult to ignite as the number of carbon atoms increases. The general equation for complete combustion is:
- CnH2n+2 + (3/2n + 1/2) O2 → (n + 1) H2O + n CO2
- or CnH2n+2 + (3n + 1/2) O2 → (n + 1) H2O + n CO2
In the absence of sufficient oxygen, carbon monoxide or even soot can be formed, as shown below:
For example, methane:
- 2 CH4 + 3 O2 → 4 H2O + 2 CO
- CH4 + O2 → 2 H2O + C
See the alkane heat of formation table for detailed data. The standard enthalpy change of combustion, ΔcH⊖, for alkanes increases by about 650 kJ/mol per CH2 group. Branched-chain alkanes have lower values of ΔcH⊖ than straight-chain alkanes of the same number of carbon atoms, and so can be seen to be somewhat more stable.
Biodegradation
[edit]Some organisms are capable of metalbolizing alkanes.[27][28] The methane monooxygenases convert methane to methanol. For higher alkanes, cytochrome P450 convert alkanes to alcohols, which are then susceptible to degradation.
Free radical reactions
[edit]Free radicals, molecules with unpaired electrons, play a large role in most reactions of alkanes. Free radical halogenation reactions occur with halogens, leading to the production of haloalkanes. The hydrogen atoms of the alkane are progressively replaced by halogen atoms. The reaction of alkanes and fluorine is highly exothermic and can lead to an explosion.[29] These reactions are an important industrial route to halogenated hydrocarbons. There are three steps:
- Initiation the halogen radicals form by homolysis. Usually, energy in the form of heat or light is required.
- Chain reaction or Propagation then takes place—the halogen radical abstracts a hydrogen from the alkane to give an alkyl radical. This reacts further.
- Chain termination where the radicals recombine.
Experiments have shown that all halogenation produces a mixture of all possible isomers, indicating that all hydrogen atoms are susceptible to reaction. The mixture produced, however, is not statistical: Secondary and tertiary hydrogen atoms are preferentially replaced due to the greater stability of secondary and tertiary free-radicals. An example can be seen in the monobromination of propane:[18]

In the Reed reaction, sulfur dioxide and chlorine convert hydrocarbons to sulfonyl chlorides under the influence of light.
Under some conditions, alkanes will undergo Nitration.
C-H activation
[edit]Certain transition metal complexes promote non-radical reactions with alkanes, resulting in so C–H bond activation reactions.[30]
Cracking
[edit]Cracking breaks larger molecules into smaller ones. This reaction requires heat and catalysts. The thermal cracking process follows a homolytic mechanism with formation of free radicals. The catalytic cracking process involves the presence of acid catalysts (usually solid acids such as silica-alumina and zeolites), which promote a heterolytic (asymmetric) breakage of bonds yielding pairs of ions of opposite charges, usually a carbocation. Carbon-localized free radicals and cations are both highly unstable and undergo processes of chain rearrangement, C–C scission in position beta (i.e., cracking) and intra- and intermolecular hydrogen transfer or hydride transfer. In both types of processes, the corresponding reactive intermediates (radicals, ions) are permanently regenerated, and thus they proceed by a self-propagating chain mechanism. The chain of reactions is eventually terminated by radical or ion recombination.[citation needed]
Isomerization and reformation
[edit]Dragan and his colleague were the first to report about isomerization in alkanes.[31] Isomerization and reformation are processes in which straight-chain alkanes are heated in the presence of a platinum catalyst. In isomerization, the alkanes become branched-chain isomers. In other words, it does not lose any carbons or hydrogens, keeping the same molecular weight.[31] In reformation, the alkanes become cycloalkanes or aromatic hydrocarbons, giving off hydrogen as a by-product. Both of these processes raise the octane number of the substance. Butane is the most common alkane that is put under the process of isomerization, as it makes many branched alkanes with high octane numbers.[31]
Other reactions
[edit]In steam reforming, alkanes react with steam in the presence of a nickel catalyst to give hydrogen and carbon monoxide.
Occurrence
[edit]Occurrence of alkanes in the Universe
[edit]

Alkanes form a small portion of the atmospheres of the outer gas planets such as Jupiter (0.1% methane, 2 ppm ethane), Saturn (0.2% methane, 5 ppm ethane), Uranus (1.99% methane, 2.5 ppm ethane) and Neptune (1.5% methane, 1.5 ppm ethane). Titan (1.6% methane), a satellite of Saturn, was examined by the Huygens probe, which indicated that Titan's atmosphere periodically rains liquid methane onto the moon's surface.[32] Also on Titan, the Cassini mission has imaged seasonal methane/ethane lakes near the polar regions of Titan. Methane and ethane have also been detected in the tail of the comet Hyakutake. Chemical analysis showed that the abundances of ethane and methane were roughly equal, which is thought to imply that its ices formed in interstellar space, away from the Sun, which would have evaporated these volatile molecules.[33] Alkanes have also been detected in meteorites such as carbonaceous chondrites.
Occurrence of alkanes on Earth
[edit]Traces of methane gas (about 0.0002% or 1745 ppb) occur in the Earth's atmosphere, produced primarily by methanogenic microorganisms, such as Archaea in the gut of ruminants.[34]
The most important commercial sources for alkanes are natural gas and oil.[18] Natural gas contains primarily methane and ethane, with some propane and butane: oil is a mixture of liquid alkanes and other hydrocarbons. These hydrocarbons were formed when marine animals and plants (zooplankton and phytoplankton) died and sank to the bottom of ancient seas and were covered with sediments in an anoxic environment and converted over many millions of years at high temperatures and high pressure to their current form. Natural gas resulted thereby for example from the following reaction:
- C6H12O6 → 3 CH4 + 3 CO2
These hydrocarbon deposits, collected in porous rocks trapped beneath impermeable cap rocks, comprise commercial oil fields. They have formed over millions of years and once exhausted cannot be readily replaced. The depletion of these hydrocarbons reserves is the basis for what is known as the energy crisis.
Alkanes have a low solubility in water, so the content in the oceans is negligible; however, at high pressures and low temperatures (such as at the bottom of the oceans), methane can co-crystallize with water to form a solid methane clathrate (methane hydrate). Although this cannot be commercially exploited at the present time, the amount of combustible energy of the known methane clathrate fields exceeds the energy content of all the natural gas and oil deposits put together. Methane extracted from methane clathrate is, therefore, a candidate for future fuels.
Biological occurrence
[edit]
Aside from petroleum and natural gas, alkanes occur significantly in nature only as methane, which is produced by some archaea by the process of methanogenesis. These organisms are found in the gut of termites[35] and cows.[36] The methane is produced from carbon dioxide or other organic compounds. Energy is released by the oxidation of hydrogen:
- CO2 + 4 H2 → CH4 + 2 H2O
It is probable that our current deposits of natural gas were formed in a similar way.[37]
Certain types of bacteria can metabolize alkanes: they prefer even-numbered carbon chains as they are easier to degrade than odd-numbered chains.[38]
Alkanes play a negligible role in higher organisms, with rare exception. Some yeasts, e.g., Candida tropicale, Pichia sp., Rhodotorula sp., can use alkanes as a source of carbon or energy. The fungus Amorphotheca resinae prefers the longer-chain alkanes in aviation fuel, and can cause serious problems for aircraft in tropical regions.[39]
In plants, the solid long-chain alkanes are found in the plant cuticle and epicuticular wax of many species, but are only rarely major constituents.[40] They protect the plant against water loss, prevent the leaching of important minerals by the rain, and protect against bacteria, fungi, and harmful insects. The carbon chains in plant alkanes are usually odd-numbered, between 27 and 33 carbon atoms in length,[40] and are made by the plants by decarboxylation of even-numbered fatty acids. The exact composition of the layer of wax is not only species-dependent but also changes with the season and such environmental factors as lighting conditions, temperature or humidity.[40]
The Jeffrey pine is noted for producing exceptionally high levels of n-heptane in its resin, for which reason its distillate was designated as the zero point for one octane rating. Floral scents have also long been known to contain volatile alkane components, and n-nonane is a significant component in the scent of some roses.[41] Emission of gaseous and volatile alkanes such as ethane, pentane, and hexane by plants has also been documented at low levels, though they are not generally considered to be a major component of biogenic air pollution.[42]
Edible vegetable oils also typically contain small fractions of biogenic alkanes with a wide spectrum of carbon numbers, mainly 8 to 35, usually peaking in the low to upper 20s, with concentrations up to dozens of milligrams per kilogram (parts per million by weight) and sometimes over a hundred for the total alkane fraction.[43]
Alkanes are found in animal products, although they are less important than unsaturated hydrocarbons. One example is the shark liver oil, which is approximately 14% pristane (2,6,10,14-tetramethylpentadecane, C19H40). They are important as pheromones, chemical messenger materials, on which insects depend for communication. In some species, e.g. the support beetle Xylotrechus colonus, pentacosane (C25H52), 3-methylpentaicosane (C26H54) and 9-methylpentaicosane (C26H54) are transferred by body contact. With others like the tsetse fly Glossina morsitans morsitans, the pheromone contains the four alkanes 2-methylheptadecane (C18H38), 17,21-dimethylheptatriacontane (C39H80), 15,19-dimethylheptatriacontane (C39H80) and 15,19,23-trimethylheptatriacontane (C40H82), and acts by smell over longer distances. Waggle-dancing honey bees produce and release two alkanes, tricosane and pentacosane.[44]
Ecological relations
[edit]
One example, in which both plant and animal alkanes play a role, is the ecological relationship between the sand bee (Andrena nigroaenea) and the early spider orchid (Ophrys sphegodes); the latter is dependent for pollination on the former. Sand bees use pheromones in order to identify a mate; in the case of A. nigroaenea, the females emit a mixture of tricosane (C23H48), pentacosane (C25H52) and heptacosane (C27H56) in the ratio 3:3:1, and males are attracted by specifically this odor. The orchid takes advantage of this mating arrangement to get the male bee to collect and disseminate its pollen; parts of its flower not only resemble the appearance of sand bees but also produce large quantities of the three alkanes in the same ratio as female sand bees. As a result, numerous males are lured to the blooms and attempt to copulate with their imaginary partner: although this endeavor is not crowned with success for the bee, it allows the orchid to transfer its pollen, which will be dispersed after the departure of the frustrated male to other blooms.
Production
[edit]Petroleum refining
[edit]
The most important source of alkanes is natural gas and crude oil.[18] Alkanes are separated in an oil refinery by fractional distillation. Unsaturated hydrocarbons are converted to alkanes by hydrogenation:[45]
- RCH=CH2 + H2 − → RCH2−CH3 (R = alkyl)
Another route to alkanes is hydrogenolysis, which entails cleavage of C-heteroatom bonds using hydrogen. In industry, the main substrates are organonitrogen and organosulfur impurities, i.e. the heteroatoms are N and S. The specific processes are called hydrodenitrification and hydrodesulfurization:
- R3N + 3 H2 → 3 RH + H3N
- R2S + 2 H2 → 2 RH + H2S
Hydrogenolysis can be applied to the conversion of virtually any functional group into hydrocarbons. Substrates include haloalkanes, alcohols, aldehydes, ketones, carboxylic acids, etc. Both hydrogenolysis and hydrogenation are practiced in refineries. The can be effected by using lithium aluminium hydride, Clemmenson reduction and other specialized routes.
Coal
[edit]Coal is a more traditional precursor to alkanes. A wide range of technologies have been intensively practiced for centuries.[45] Simply heating coal gives alkanes, leaving behind coke. Relevant technologies include the Bergius process and coal liquifaction. Partial combustion of coal and related solid organic compounds generates carbon monoxide, which can be hydrogenated using the Fischer–Tropsch process. This technology allows the synthesize liquid hydrocarbons, including alkanes. This method is used to produce substitutes for petroleum distillates.
Laboratory preparation
[edit]Rarely is there any interest in the synthesis of alkanes, since they are usually commercially available and less valued than virtually any precursor. The best-known method is hydrogenation of alkenes. Many C-X bonds can be converted to C-H bonds using lithium aluminium hydride, Clemmenson reduction, and other specialized routes.[46] Hydrolysis of Alkyl Grignard reagents and alkyl lithium compounds gives alkanes.[47]
Applications
[edit]Fuels
[edit]The dominant use of alkanes is as fuels. Propane and butane, easily liquified gases, are commonly known as liquified petroleum gas (LPG).[48] From pentane to octane the alkanes are highly volatile liquids. They are used as fuels in internal combustion engines, as they vaporize easily on entry into the combustion chamber without forming droplets, which would impair the uniformity of the combustion. Branched-chain alkanes are preferred as they are much less prone to premature ignition, which causes knocking, than their straight-chain homologues. This propensity to premature ignition is measured by the octane rating of the fuel, where 2,2,4-trimethylpentane (isooctane) has an arbitrary value of 100, and heptane has a value of zero. Apart from their use as fuels, the middle alkanes are also good solvents for nonpolar substances. Alkanes from nonane to, for instance, hexadecane (an alkane with sixteen carbon atoms) are liquids of higher viscosity, less and less suitable for use in gasoline. They form instead the major part of diesel and aviation fuel. Diesel fuels are characterized by their cetane number, cetane being an old name for hexadecane. However, the higher melting points of these alkanes can cause problems at low temperatures and in polar regions, where the fuel becomes too thick to flow correctly.
Precursors to chemicals
[edit]By the process of cracking, alkanes can be converted to alkenes. Simple alkenes are precursors to polymers, such as polyethylene and polypropylene. When the cracking is taken to extremes, alkanes can be converted to carbon black, which is a significant tire component.
Chlorination of methane gives chloromethanes, which are used as solvents and building blocks for complex compounds. Similarly treatment of methane with sulfur gives carbon disulfide. Still other chemicals are prepared by reaction with sulfur trioxide and nitric oxide
Other
[edit]Some light hydrocarbons are used as aerosol sprays.
Alkanes from hexadecane upwards form the most important components of fuel oil and lubricating oil. In the latter function, they work at the same time as anti-corrosive agents, as their hydrophobic nature means that water cannot reach the metal surface. Many solid alkanes find use as paraffin wax, for example, in candles. This should not be confused however with true wax, which consists primarily of esters.
Alkanes with a chain length of approximately 35 or more carbon atoms are found in bitumen, used, for example, in road surfacing. However, the higher alkanes have little value and are usually split into lower alkanes by cracking.
Hazards
[edit]Alkanes are highly flammable, but they have low toxicities. Methane "is toxicologically virtually inert." Alkanes can be asphyxiants and narcotic.[45]
See also
[edit]References
[edit]- ^ a b IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "alkanes". doi:10.1351/goldbook.A00222
- ^ Smith, Michael B.; March, Jerry (2007). Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.). New York: Wiley-Interscience. p. 23. ISBN 978-0-471-72091-1.
- ^ On-Line Encyclopedia of Integer Sequences (sequence A000602 in the OEIS) Number of n-node unrooted quartic trees; number of n-carbon alkanes C(n)H(2n+2) ignoring stereoisomers
- ^ Alabugin, Igor V. (2016). Stereoelectronic effects : a bridge between structure and reactivity. Wiley. ISBN 978-1-118-90637-8. OCLC 957525299.
- ^ IUPAC, Commission on Nomenclature of Organic Chemistry (1993). "R-2.2.1: Hydrocarbons". A Guide to IUPAC Nomenclature of Organic Compounds (Recommendations 1993). Blackwell Scientific. ISBN 978-0-632-03488-8. Retrieved 12 February 2007.
- ^ "Alkane Nomenclature". Archived from the original on 2 February 2012.
- ^ Thus, the ending "-diene" is applied in some cases where von Hofmann had "-ine"
- ^ a b Reusch, William. "Nomenclature – Alkanes". Virtual Textbook of Organic Chemistry. Archived from the original on 21 May 2016. Retrieved 5 April 2007.
- ^ Reusch, William. "Examples of the IUPAC Rules in Practice". Virtual Textbook of Organic Chemistry. Archived from the original on 21 May 2016. Retrieved 5 April 2007.
- ^ "IUPAC Rules". www.chem.uiuc.edu. Retrieved 13 August 2018.
- ^ "Definition of CYCLOALKANES". www.merriam-webster.com. Retrieved 26 June 2021.
- ^ "Definition of NAPHTHENES". www.merriam-webster.com. Retrieved 26 June 2021.
- ^ Mackay, Donald (14 March 2006). Handbook of Physical-Chemical Properties and Environmental Fate for Organic Chemicals. CRC Press. p. 206. ISBN 1-4200-4439-7.
- ^ "Pharmaceutical Chemistry" (PDF). Archived from the original (PDF) on 29 October 2013. Retrieved 17 February 2014.
- ^ "13. Hydrocarbons | Textbooks". textbook.s-anand.net. Archived from the original on 8 May 2011. Retrieved 3 October 2014.
- ^ "Molecule Gallery - Alkanes". www.angelo.edu. Retrieved 6 December 2021.
- ^ Allaby, Michael, ed. (1988). "Alkanes (paraffins)". Illustrated Dictionary of Science, Andromeda. Windmill Books (Andromeda International).
- ^ a b c d e f R. T. Morrison; R. N. Boyd (1992). Organic Chemistry (6th ed.). Prentice Hall. ISBN 978-0-13-643669-0.
- ^ Boese, Roland; Weiss, Hans-Christoph; Bläser, Dieter (1 April 1999). "The Melting Point Alternation in the Short-Chain n-Alkanes: Single-Crystal X-Ray Analyses of Propane at 30 K and of n-Butane to n-Nonane at 90 K". Angewandte Chemie International Edition. 38 (7): 988–992. doi:10.1002/(SICI)1521-3773(19990401)38:7<988::AID-ANIE988>3.0.CO;2-0. ISSN 1433-7851.
- ^ "Solid methane". Visualization of Molecules and Crystal Structures.
- ^ Ouellette, Robert J.; Rawn, J. David (1 January 2015). "Alkanes and Cycloalkanes". Principles of Organic Chemistry. Elsevier. pp. 65–94. doi:10.1016/b978-0-12-802444-7.00003-3. ISBN 978-0-12-802444-7.
- ^ Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, p. 195, ISBN 978-0-471-72091-1
- ^ Silverstein, Robert M.; Webster, Francis X.; Kiemle, David J.; Bryce, David L. (2016). Spectrometric Identification of Organic Compounds (8th ed.). Wiley. ISBN 978-0-470-61637-6.
- ^ "Dodecane: IR Spectrum". NIST Chemistry WebBook. SRD 69.
- ^ "Dodecane". NIST Chemistry WebBook. SRD 69.
- ^ Olah, G.A.; Schlosberg, R.H. (1968). "Chemistry in Super Acids. I. Hydrogen Exchange and Polycondensation of Methane and Alkanes in FSO3H–SbF5 ("Magic Acid") Solution. Protonation of Alkanes and the Intermediacy of CH5+ and Related Hydrocarbon Ions. The High Chemical Reactivity of "Paraffins" in Ionic Solution Reactions". Journal of the American Chemical Society. 90 (10): 2726–7. doi:10.1021/ja01012a066.
- ^ Ji, Yurui; Mao, Guannan; Wang, Yingying; Bartlam, Mark (2013). "Structural insights into diversity and n-alkane biodegradation mechanisms of alkane hydroxylases". Frontiers in Microbiology. 4: 58. doi:10.3389/fmicb.2013.00058. PMC 3604635. PMID 23519435.
- ^ Dashti, Narjes; Ali, Nedaa; Eliyas, Mohamed; Khanafer, Majida; Sorkhoh, Naser A.; Radwan, Samir S. (March 2015). "Most Hydrocarbonoclastic Bacteria in the Total Environment are Diazotrophic, which Highlights Their Value in the Bioremediation of Hydrocarbon Contaminants". Microbes and Environments. 30 (1): 70–75. doi:10.1264/jsme2.ME14090. ISSN 1342-6311. PMC 4356466. PMID 25740314.
- ^ Siegemund, Günter; Schwertfeger, Werner; Feiring, Andrew; Smart, Bruce; Behr, Fred; Vogel, Herward; McKusick, Blaine. "Fluorine Compounds, Organic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a11_349. ISBN 978-3527306732.
- ^ Yu, Isaac F.; Wilson, Jake W.; Hartwig, John F. (2023). "Transition-Metal-Catalyzed Silylation and Borylation of C–H Bonds for the Synthesis and Functionalization of Complex Molecules". Chemical Reviews. 123 (19): 11619–63. doi:10.1021/acs.chemrev.3c00207. PMID 37751601. S2CID 263150991.
- ^ a b c Asinger, Friedrich (1967). Paraffins; Chemistry and Technology. Pergamon Press. OCLC 556032.
- ^ Lakdawalla, Emily. "Titan: Arizona in an Icebox?". Archived from the original on 6 April 2008. Retrieved 21 January 2004.
- ^ Mumma, M.J.; Disanti, M.A.; dello Russo, N.; Fomenkova, M.; Magee-Sauer, K.; Kaminski, C.D.; D.X., Xie (1996). "Detection of Abundant Ethane and Methane, Along with Carbon Monoxide and Water, in Comet C/1996 B2 Hyakutake: Evidence for Interstellar Origin". Science. 272 (5266): 1310–4. Bibcode:1996Sci...272.1310M. doi:10.1126/science.272.5266.1310. PMID 8650540. S2CID 27362518.
- ^ Janssen, P. H.; Kirs, M. (2008). "Structure of the Archaeal Community of the Rumen". Appl Environ Microbiol. 74 (12): 3619–25. Bibcode:2008ApEnM..74.3619J. doi:10.1128/AEM.02812-07. PMC 2446570. PMID 18424540.
- ^ Buczkowski, Grzegorz; Bertelsmeier, Cleo (15 January 2017). "Invasive termites in a changing climate: A global perspective". Ecology and Evolution. 7 (3): 974–985. Bibcode:2017EcoEv...7..974B. doi:10.1002/ece3.2674. PMC 5288252. PMID 28168033.
- ^ Blitz, Matt. "Do Cow Farts Actually Contribute to Global Warming?". TodayIFoundOut.com. Retrieved 11 April 2018 – via Gizmodo.
- ^ "Natural Gas". Resources Library. National Geographic Society. Retrieved 11 April 2018.
- ^ "Metabolism of Alkanes and Fatty Acids – eQuilibrator 0.2 beta documentation". equilibrator.weizmann.ac.il. Retrieved 11 April 2018.
- ^ Hendey, N. I. (1964). "Some observations on Cladosporium resinae as a fuel contaminant and its possible role in the corrosion of aluminium alloy fuel tanks". Transactions of the British Mycological Society. 47 (7): 467–475. doi:10.1016/s0007-1536(64)80024-3.
- ^ a b c Baker, E.A. (1982). "Chemistry and morphology of plant epicuticular waxes". In Cutler, D.F.; Alvin, K.L.; Price, C.E. (eds.). The Plant Cuticle. Academic Press. pp. 139–165. ISBN 0-12-199920-3.
- ^ Kim, HyunJung; Kim, NamSun; Lee, DongSun (2000). "Determination of floral fragrances of Rosa hybrida using solid-phase trapping-solvent extraction and gas chromatography–mass spectrometry". Journal of Chromatography A. 902 (2): 389–404. doi:10.1016/S0021-9673(00)00863-3. PMID 11192171.
- ^ Kesselmeier, J.; Staudt, N. (1999). "Biogenic Volatile Organic Compounds (VOC): An Overview on Emission, Physiology and Ecology" (PDF). Journal of Atmospheric Chemistry. 33 (1): 22–38. Bibcode:1999JAtC...33...23K. doi:10.1023/A:1006127516791. S2CID 94021819. Archived from the original (PDF) on 13 March 2013.
- ^ Moreda, W.; Perez-Camino, M. C.; Cert, A. (2001). "Gas and liquid chromatography of hydrocarbons in edible vegetable oils". Journal of Chromatography A. 936 (1–2): 159–171. doi:10.1016/s0021-9673(01)01222-5. PMID 11760997.
- ^ Thom C, Gilley DC, Hooper J, Esch HE (21 August 2007). "The Scent of the Waggle Dance". PLOS Biology. 5 (9): e228. doi:10.1371/journal.pbio.0050228. PMC 1994260. PMID 17713987.
- ^ a b c Schmidt, Roland; Griesbaum, Karl; Behr, Arno; Biedenkapp, Dieter; Voges, Heinz-Werner; Garbe, Dorothea; Paetz, Christian; Collin, Gerd; Mayer, Dieter; Höke, Hartmut. "Hydrocarbons". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a13_227.pub3. ISBN 978-3527306732.
- ^ Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, p. 1790, ISBN 978-0-471-72091-1
- ^ Noller, C.R. (1931). "n-Pentane". Organic Syntheses. 11: 84. doi:10.15227/orgsyn.011.0084.
- ^ "Using propane as a fuel" (PDF). Archived from the original (PDF) on 12 October 2013. Retrieved 27 November 2012.
Further reading
[edit]- Virtual Textbook of Organic Chemistry
- A visualization of the crystal structures of alkanes up to nonan
- Redwood, Boverton (1911). . Encyclopædia Britannica. Vol. 20 (11th ed.). pp. 752–756.


